|
PromoCell
placental pericytes 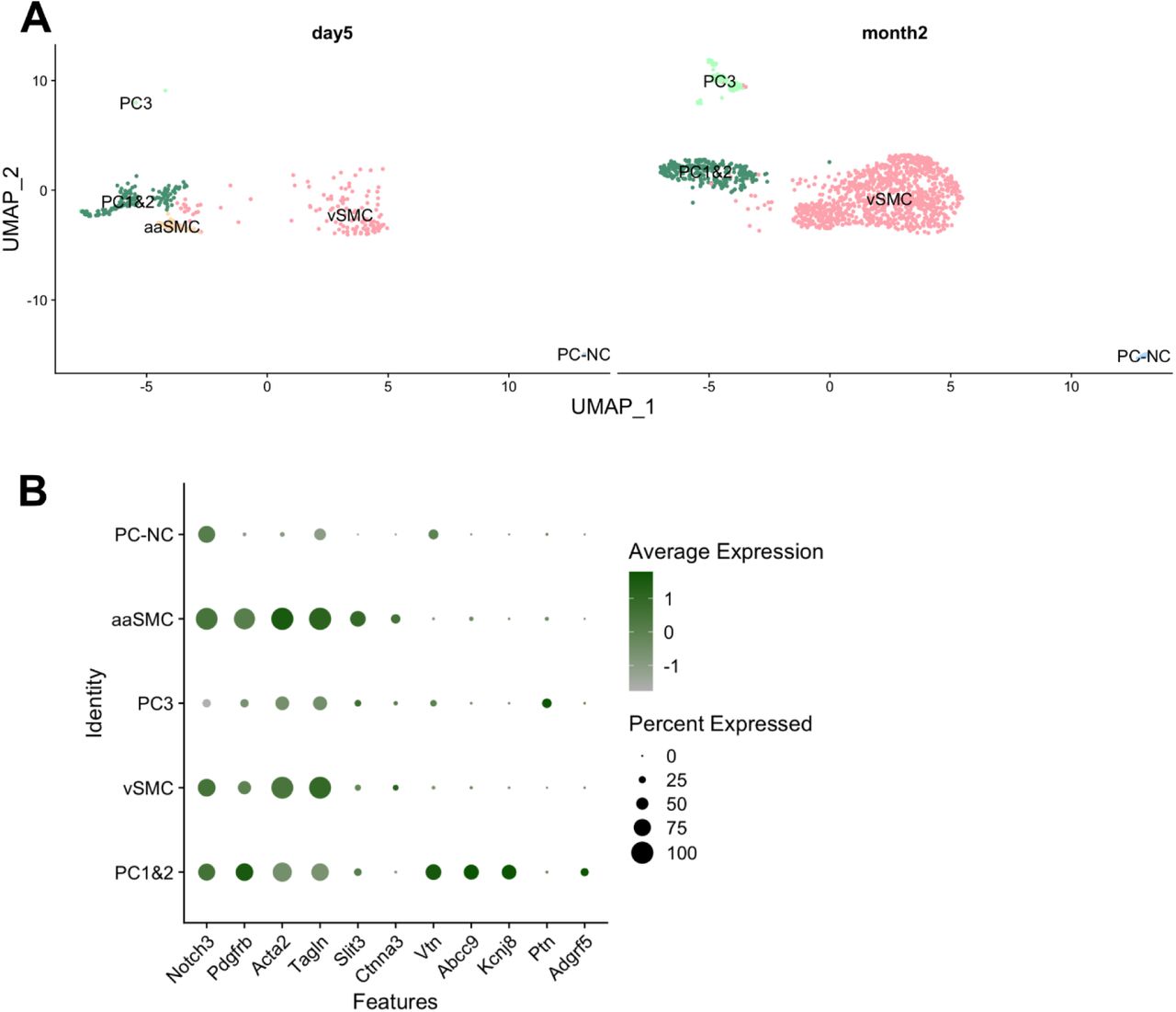 Placental Pericytes, supplied by PromoCell, used in various techniques. Bioz Stars score: 95/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/placental pericytes/product/PromoCell Average 95 stars, based on 1 article reviews
placental pericytes - by Bioz Stars,
2026-03
95/100 stars
|
Buy from Supplier |
|
iXCells Biotechnologies
human brain vascular pericytes  Human Brain Vascular Pericytes, supplied by iXCells Biotechnologies, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/human brain vascular pericytes/product/iXCells Biotechnologies Average 94 stars, based on 1 article reviews
human brain vascular pericytes - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
|
PromoCell
pericyte growth medium  Pericyte Growth Medium, supplied by PromoCell, used in various techniques. Bioz Stars score: 95/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/pericyte growth medium/product/PromoCell Average 95 stars, based on 1 article reviews
pericyte growth medium - by Bioz Stars,
2026-03
95/100 stars
|
Buy from Supplier |
|
Angio-Proteomie
htert  Htert, supplied by Angio-Proteomie, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/htert/product/Angio-Proteomie Average 93 stars, based on 1 article reviews
htert - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
PromoCell
primary human placental pericytes  Primary Human Placental Pericytes, supplied by PromoCell, used in various techniques. Bioz Stars score: 95/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/primary human placental pericytes/product/PromoCell Average 95 stars, based on 1 article reviews
primary human placental pericytes - by Bioz Stars,
2026-03
95/100 stars
|
Buy from Supplier |
|
Angio-Proteomie
pericyte growth medium  Pericyte Growth Medium, supplied by Angio-Proteomie, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/pericyte growth medium/product/Angio-Proteomie Average 93 stars, based on 1 article reviews
pericyte growth medium - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
Innoprot Inc
pericyte medium pm  Pericyte Medium Pm, supplied by Innoprot Inc, used in various techniques. Bioz Stars score: 92/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/pericyte medium pm/product/Innoprot Inc Average 92 stars, based on 1 article reviews
pericyte medium pm - by Bioz Stars,
2026-03
92/100 stars
|
Buy from Supplier |
|
Angio-Proteomie
human brain pericytes  Human Brain Pericytes, supplied by Angio-Proteomie, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/human brain pericytes/product/Angio-Proteomie Average 94 stars, based on 1 article reviews
human brain pericytes - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
|
Angio-Proteomie
human placental microvascular pericytes  Human Placental Microvascular Pericytes, supplied by Angio-Proteomie, used in various techniques. Bioz Stars score: 88/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/human placental microvascular pericytes/product/Angio-Proteomie Average 88 stars, based on 1 article reviews
human placental microvascular pericytes - by Bioz Stars,
2026-03
88/100 stars
|
Buy from Supplier |
|
iXCells Biotechnologies
mouse brain vascular  Mouse Brain Vascular, supplied by iXCells Biotechnologies, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/mouse brain vascular/product/iXCells Biotechnologies Average 94 stars, based on 1 article reviews
mouse brain vascular - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
|
Angio-Proteomie
placental microvascular pericytes  Placental Microvascular Pericytes, supplied by Angio-Proteomie, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/placental microvascular pericytes/product/Angio-Proteomie Average 93 stars, based on 1 article reviews
placental microvascular pericytes - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
iXCells Biotechnologies
human pericyte growth medium 5  Human Pericyte Growth Medium 5, supplied by iXCells Biotechnologies, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/human pericyte growth medium 5/product/iXCells Biotechnologies Average 94 stars, based on 1 article reviews
human pericyte growth medium 5 - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
Image Search Results

Journal: bioRxiv
Article Title: A role for fibroblast and mural cell subsets in models of neuropathic pain
doi: 10.1101/2024.12.11.627455
Figure Lengend Snippet: Mouse mural cells sub-cluster into five groups, resembling cell types previously described in the brain, with one (aaSMC) detected only day 5 after PSNL or sham surgery (A). Acta2 & Tagln high vascular smooth muscle cells (vSMC), arteriolar SMCs enriched for Slit3 and Ctnna3 (aaSMC), Vtn & Abcc9 & Kcnj8 positive pericytes (PC1&2), Adgrf5 negative (PC3) and Acta2 negative pericytes (PC-NC). Expression of marker genes is plotted in B. No prominent differences were detected between sham and PSNL groups at either timepoints.
Article Snippet: A vial of human
Techniques: Expressing, Marker
Journal: bioRxiv
Article Title: A role for fibroblast and mural cell subsets in models of neuropathic pain
doi: 10.1101/2024.12.11.627455
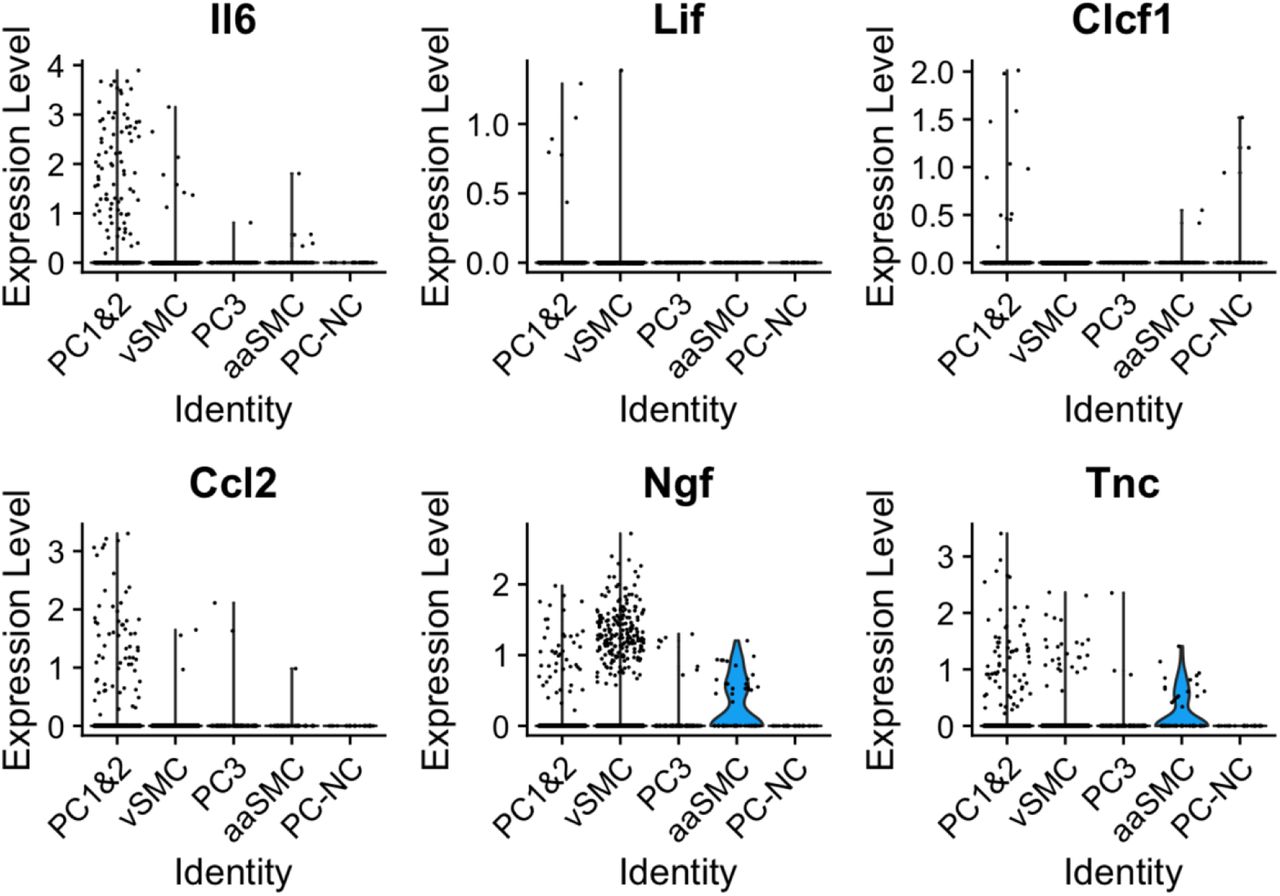
Figure Lengend Snippet: Plotted here is a selection of mediators, the majority of which we identified as differentially expressed in pericytes in our bulk sequencing experiment (Il6, Lif, Clcf1, Ccl2 and Tnc). Their expression patterns appeared to differ across mural cell populations, with significant enrichment of Il6, Ccl2 and Tnc in PC1&2 pericytes, and Clcf1 in Acta2 negative pericytes (PC-NC).
Article Snippet: A vial of human
Techniques: Selection, Sequencing, Expressing
Journal: bioRxiv
Article Title: A role for fibroblast and mural cell subsets in models of neuropathic pain
doi: 10.1101/2024.12.11.627455
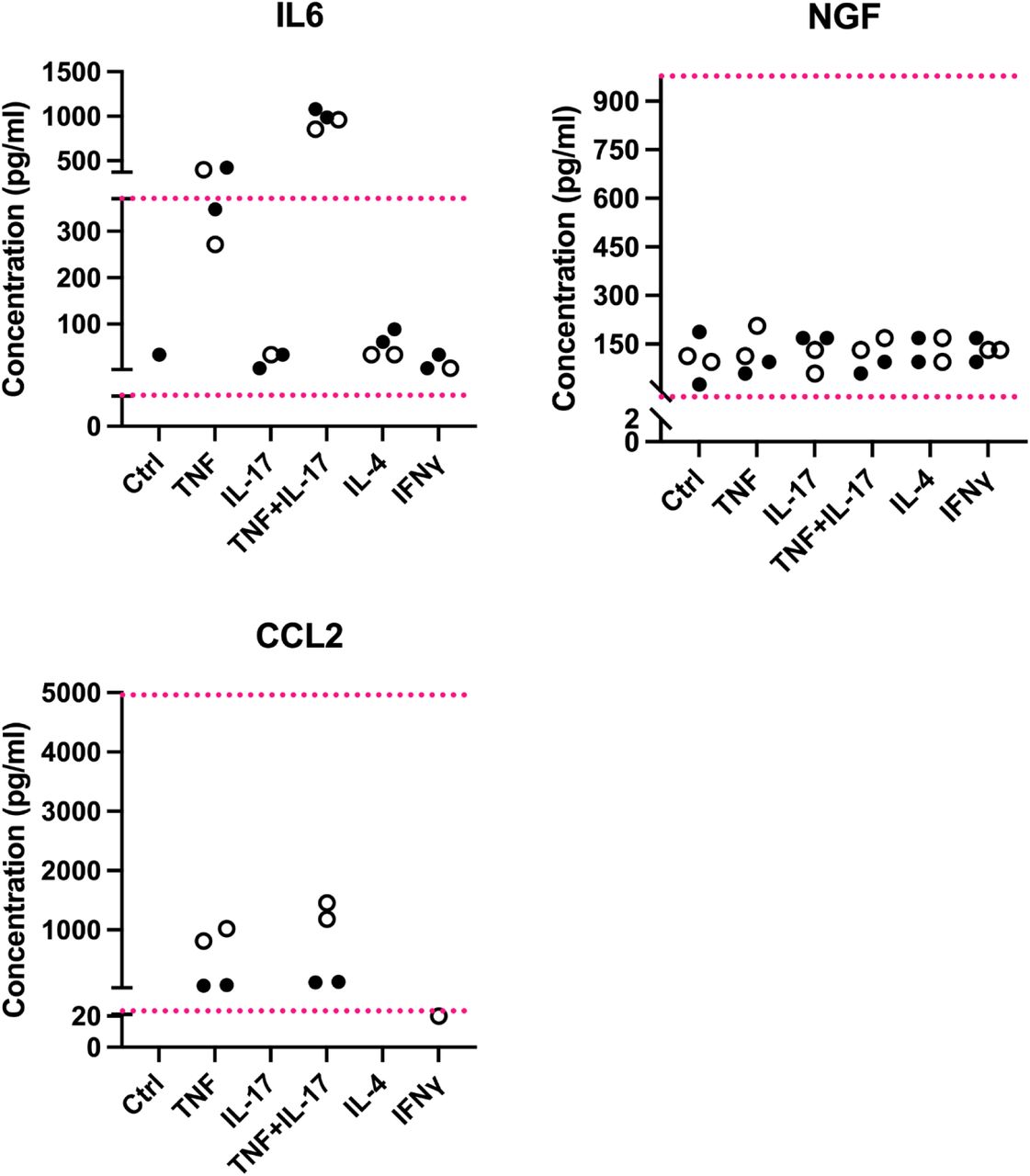
Figure Lengend Snippet: Each dot is a well of cells. Filled circles shows the expression levels obtained from media taken from a placental pericyte line, open circles those obtained from a nerve pericyte line. Ctrl = regular pericyte media; other columns: pericytes treated for four hours with TNF (10ng/ml), IL-17 (10ng/ml), TNF+IL-17 (both at 10ng/ml), IL-4 (10ng/ml) or IFNγ (100ng/ml). The red dotted lines represent the upper and lower limits of the standard curves for each protein, i.e. the upper and lower detection limits. Shown here are 1:100 dilutions for IL-6 and NGF and 1:2 dilutions for CCL2.
Article Snippet: A vial of human
Techniques: Expressing
Journal: bioRxiv
Article Title: A role for fibroblast and mural cell subsets in models of neuropathic pain
doi: 10.1101/2024.12.11.627455
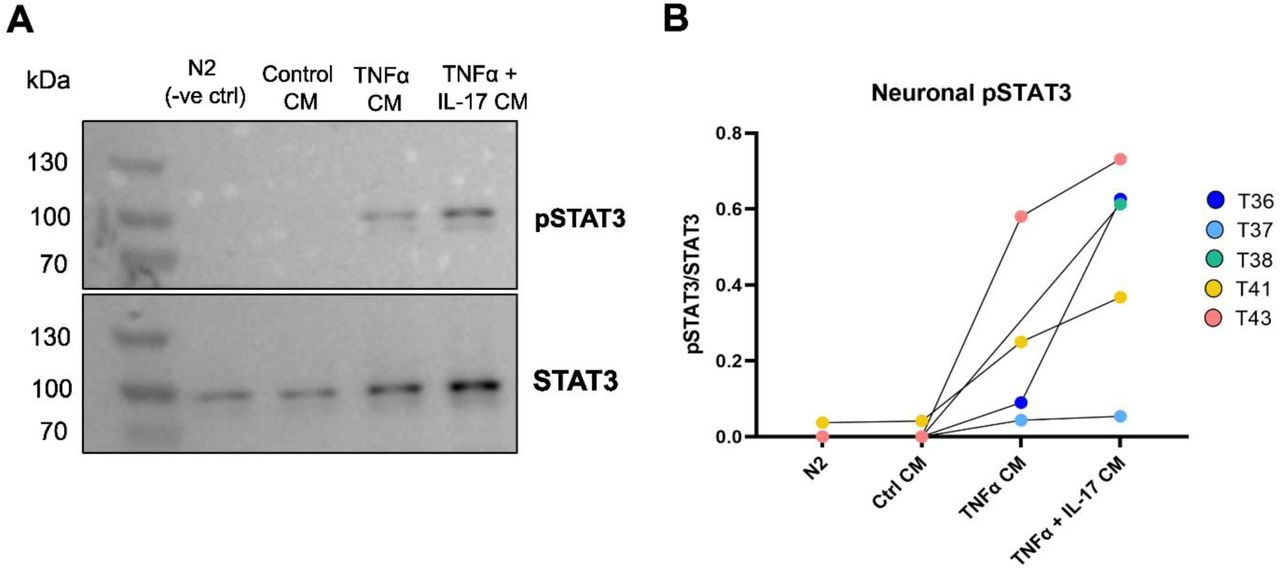
Figure Lengend Snippet: (A) Representative Western blot of neuronal pSTAT3 and STAT3 following conditioned media incubation. See Supplementary Materials for all full-length Western Blots. (B) Quantification of neuronal pSTAT3/STAT3 following incubation with cytokine-activated pericyte conditioned media (CM). Each dot represents an independent neuron differentiation (T36-43, n=5). N2 = neuronal control medium.
Article Snippet: A vial of human
Techniques: Western Blot, Incubation, Control

Journal: Advanced Science
Article Title: Pericytes Contribute to Dysfunction in a Human 3D Model of Placental Microvasculature through VEGF‐Ang‐Tie2 Signaling
doi: 10.1002/advs.201900878
Figure Lengend Snippet: Placental pericytes reduce microvessel growth and connectivity. a) Schematic diagram showing pericyte location in relation to microvessels in vivo. b) Confocal image of HPP‐cocultured with HUVEC fixed at day 5. Shown is a single XY plane and orthogonal projections demonstrating lumen (red) wrapped by HPPs (green), as indicated by white arrows. Nuclei were labeled with Dapi (blue). Scale bar is 200 µm. c) Schematic showing the various geometric measurements using binary projection images. d) Comparison of mean vessel area (EC coverage), branch length, and microvessel connectivity between HLF and HPP cocultures. Significant differences between parameters appear early on. e) Parameters are compared for HPP cocultures with (green) and without (gray) added VEGF+FGF. Shown is mean ± s.e.m. * P > 0.05 with t ‐test.
Article Snippet: GFP‐labeled HPP (microvascular) were acquired from
Techniques: In Vivo, Labeling, Comparison

Journal: Advanced Science
Article Title: Pericytes Contribute to Dysfunction in a Human 3D Model of Placental Microvasculature through VEGF‐Ang‐Tie2 Signaling
doi: 10.1002/advs.201900878
Figure Lengend Snippet: A triculture model for increased microvessel connectivity. a) A triculture microvascular system perfused with fluorescently labeled beads. HUVEC—red, pericytes—green, 10 µm beads—magenta. b) Binary images from maximum intensity projections for co‐ and tricultures, as shown at day 5. c) Schedule for media change from full growth endothelial growth medium (EGM) to reduced serum basal medium (EBM). d) Representative flow cytometry density plots for HPP, HLF, and tricultures. e) Mean population of ECs and stromal cells for the co‐ and tricultures at day 5, as measured by flow cytometry. Three separate devices for each culture condition were used for measurement and repeated in n = 3 separate experiments. f) Microvessel parameters are compared between co‐ and tricultures. Shown is mean ± s.e.m. Significance is indicated by * P < 0.05, ** P < 0.01, *** P < 0.001, **** P < 0.0001, one‐way ANOVA and Tukey test.
Article Snippet: GFP‐labeled HPP (microvascular) were acquired from
Techniques: Labeling, Flow Cytometry

Journal: Advanced Science
Article Title: Pericytes Contribute to Dysfunction in a Human 3D Model of Placental Microvasculature through VEGF‐Ang‐Tie2 Signaling
doi: 10.1002/advs.201900878
Figure Lengend Snippet: Pericytes influence PE‐affiliated cytokine expression and endothelial barrier function. a) Cytokine expression is shown for HPP, HLF, and triculture microvessel supernatants collected at day 5. HPPs result in increased PE‐associated cytokine expression, as indicated by the last row demonstrating those that are up (+) and down (−) regulated in PE. ND—no‐data in found. Here red values are high, blue low, and white are mid‐level (0.5). All cytokines were normalized to numbers between 1 and 0 based on maximum and minimum intensities from the cytokine array (Figure S4b, Supporting Information). b) Ang1/2 expression analyzed by ELISA for co‐ and tricultures, measured from pooled samples ( n = 5). c) Permeability of microvessels perfused with 10 kDa dextran (blue) at day 7 for co‐ and tricultures. Shown is mean ± s.e.m. Significance is indicated by * P < 0.05, ** P < 0.01, using t ‐test. d) Confocal images demonstrating perfusability of HLF cocultures and Tricultures, and lack of perfusability in HPP cocultures. HUVEC—red, 10 kDa dextran–blue. Scale bar is 200 µm.
Article Snippet: GFP‐labeled HPP (microvascular) were acquired from
Techniques: Expressing, Enzyme-linked Immunosorbent Assay, Permeability

Journal: International Journal of Molecular Sciences
Article Title: Effects of High Glucose Concentration on Pericyte-Like Differentiated Human Adipose-Derived Mesenchymal Stem Cells
doi: 10.3390/ijms22094604
Figure Lengend Snippet: Representative photomicrographs of human adipose mesenchymal stem cells cultured in basal medium (ASC) or in pericyte medium (pmASC), stained by crystal violet during cellular proliferation assays. After three days of growth in their respective medium, glucose was added in some samples (High Glucose, 25 mM, HG), whereas other samples were kept in normal glucose (NG) condition. Pictures were taken after 72 h from glucose addition and compared to NG samples. A : ASC NG; B : pmASC NG; C : ASC HG; D : pmASC HG. In each image, two magnification levels are shown (10× and 4×, respectively). Scale bar: 100 µm.
Article Snippet: HRPCs were seeded in poly-L-lysine (0.01 mg/mL solution, Innoprot) coated culture flasks and incubated at 37 °C with 5% CO 2 in a specific
Techniques: Cell Culture, Staining

Journal: International Journal of Molecular Sciences
Article Title: Effects of High Glucose Concentration on Pericyte-Like Differentiated Human Adipose-Derived Mesenchymal Stem Cells
doi: 10.3390/ijms22094604
Figure Lengend Snippet: High glucose effects on cell proliferation and viability in human adipose mesenchymal stem cells cultured in basal medium (ASC) or in pericyte medium (pmASC), and in human retinal pericytes (HRPC). In some samples, glucose was added to the culture medium (High Glucose, 25 mM, HG); data were gathered after 24 h, 48 h and 72 h from glucose addition and compared to corresponding samples kept in normal glucose (NG) condition. Proliferation rate was assessed by crystal violet assays in ASC, pmASC ( A ), and HRPC cultures ( B ) in NG or in HG. Cell viability was evaluated by MTT assays in ASC, pmASC ( C ), and HRPC cultures ( D ) in NG or in HG. Absorbance values were determined at 570 nm for both assays. Values are expressed as mean ± SEM of three independent experiments. In A and C, values are referred to ASC NG population, at each corresponding time point. In B and D, values are referred to HRPC NG population, at each corresponding time point. * p < 0.05 pmASC vs. ASC; # p < 0.05 HG vs. NG. Two-way ANOVA, followed by Sidak’s test.
Article Snippet: HRPCs were seeded in poly-L-lysine (0.01 mg/mL solution, Innoprot) coated culture flasks and incubated at 37 °C with 5% CO 2 in a specific
Techniques: Cell Culture

Journal: International Journal of Molecular Sciences
Article Title: Effects of High Glucose Concentration on Pericyte-Like Differentiated Human Adipose-Derived Mesenchymal Stem Cells
doi: 10.3390/ijms22094604
Figure Lengend Snippet: Cell migration ability evaluated by wound-healing assays in human adipose mesenchymal stem cells cultured in basal medium (ASC) or in pericyte medium (pmASC), and in cultures of human retinal pericytes (HRPC). In some samples of each culture type, glucose was added to the culture medium (High Glucose, 25 mM, HG) whereas other samples were kept in normal glucose (NG) condition. ( A ): Representative pictures of each sample taken immediately after the scratch (0 h), after 24 h and 48 h of culture. Scale bar: 100µm (Magnification: 4×). Percentage of wound closure was quantified by Image J software for ASC and pm ASC cultures ( B ), and HRPC cultures ( C ). Values are expressed as mean ± SEM of three independent experiments. * p < 0.05 pmASC vs. ASC; # p < 0.05 HG vs. NG. Two-way ANOVA, followed by Sidak’s test.
Article Snippet: HRPCs were seeded in poly-L-lysine (0.01 mg/mL solution, Innoprot) coated culture flasks and incubated at 37 °C with 5% CO 2 in a specific
Techniques: Migration, Cell Culture, Software

Journal: International Journal of Molecular Sciences
Article Title: Effects of High Glucose Concentration on Pericyte-Like Differentiated Human Adipose-Derived Mesenchymal Stem Cells
doi: 10.3390/ijms22094604
Figure Lengend Snippet: High glucose effects on ROS levels evaluated by H2DCFDA assays in ( A ) human adipose mesenchymal stem cells cultured in basal medium (ASC) or in pericyte medium (pmASC), and in ( B ) human retinal pericytes (HRPC). In some samples, glucose was added to the culture medium (High Glucose, 25 mM, HG); data were gathered after 24 h, 48 h and 72 h from glucose addition, and compared to samples kept in normal glucose (NG) condition. ROS values of each group are normalized by the corresponding population density, as determined by crystal violet staining, at each corresponding time point (ROS/CV ratio). Values are expressed as mean ± SEM of three independent experiments. * p < 0.05 pmASC vs. ASC; # p < 0.05 HG vs. NG; ‡ p < 0.05 72 h vs. 24 h; Two-way ANOVA, followed by Sidak’s test.
Article Snippet: HRPCs were seeded in poly-L-lysine (0.01 mg/mL solution, Innoprot) coated culture flasks and incubated at 37 °C with 5% CO 2 in a specific
Techniques: Cell Culture, Staining

Journal: International Journal of Molecular Sciences
Article Title: Effects of High Glucose Concentration on Pericyte-Like Differentiated Human Adipose-Derived Mesenchymal Stem Cells
doi: 10.3390/ijms22094604
Figure Lengend Snippet: High glucose effects on anti- and pro-inflammatory cytokines and angiogenic factors in human adipose mesenchymal stem cells cultured in basal medium (ASC) or in pericyte medium (pmASC). In some samples, glucose was added to the culture medium (High Glucose, 25 mM, HG); data were gathered after 72 h from glucose addition and compared to corresponding samples kept in normal glucose (NG) condition. Quantitative analysis of IL 10 ( A ), TGF-β1 ( B ), TNFα ( C ), IL-1β ( D ), VEGF ( E ), Angiopoietin-2 ( F ) and MMP9 ( G ) mRNA levels, as evaluated by qRT-PCR. mRNA levels of each group were normalized to the housekeeping reference gene ribosomal 18S RNA. In the histograms, values are expressed as a fold change of those detected in ASCs cultured in NG condition. Each value represents mean ± SEM obtained from three independent experiments. * p < 0.05 pmASC vs. ASC; # p < 0.05 HG vs. NG. Two-way ANOVA, followed by Sidak’s test.
Article Snippet: HRPCs were seeded in poly-L-lysine (0.01 mg/mL solution, Innoprot) coated culture flasks and incubated at 37 °C with 5% CO 2 in a specific
Techniques: Cell Culture, Quantitative RT-PCR