|
Carna Inc
gst Gst, supplied by Carna Inc, used in various techniques. Bioz Stars score: 96/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/gst/product/Carna Inc Average 96 stars, based on 1 article reviews
gst - by Bioz Stars,
2026-04
96/100 stars
|
Buy from Supplier |
|
StressMarq
p wnk1 ps382 antibody ![( A ) Genotyping of <t>Wnk1</t> -cKO mice mediated by neuron-specific Syn1-Cre . Genomic tail-clip DNA was used for analysis. Lane 1, Wnk1 fl/+ ; Syn1-Cre ; lane 2, Wnk1 fl/fl ; lane 3, Wnk1 fl/fl ; Syn1-Cre . PCR is shown to detect WT vs. Wnk1 fl/fl locus (exon 2 and neo cassette are floxed). PCR forward primer F is located at exon 2. Reverse primers R1 and R2 are located at intron 2 and neo cassette, respectively. Note that Syn1-Cre is only active in neurons, so unexcised Wnk1 fl/fl locus is detected in tail-clip DNA. With large size neo cassette in the floxed locus, the F/R1 primer set does not amplify under the condition of PCR reaction. Additionally, PCR is shown to detect Syn1-Cre using Cre-specific primers. ( B ) Representative Western blot of WNK1 protein in WT and cKO brain regions shown relative to the kidney. Hippo, hippocampus. Cortex, cerebral cortex. ( C ) Quantitation (mean ± SEM) of 4 separate experiments, as shown in B . One WT and cKO mouse for each experiment. WNK1 was normalized to Gapdh and compared with the WT kidney (set as “1”). * P < 0.05, # P < 0.01, KO vs. WT by unpaired t test. ( D ) Atlas of brain section for immunofluorescent staining, as in E and F . OVLT (also known as vascular-organ-of-lamina-terminalis [VOLT]) is marked by a red line. 3V, third ventricle; MnPO, median preoptic nucleus; MPA, medial preoptic area. ( E and F ) Immunofluorescent staining of WNK1 in OVLT neurons colocalized with neuronal marker β-3 tubulin in WT ( E ) and cKO ( F ) mice. Scale bar: 100 μm.](https://pub-med-central-images-cdn.bioz.com/pub_med_central_ids_ending_with_1991/pmc10231991/pmc10231991__jci-133-164222-g062.jpg) P Wnk1 Ps382 Antibody, supplied by StressMarq, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/p wnk1 ps382 antibody/product/StressMarq Average 93 stars, based on 1 article reviews
p wnk1 ps382 antibody - by Bioz Stars,
2026-04
93/100 stars
|
Buy from Supplier |
|
OriGene
recombinant full length wnk1  Recombinant Full Length Wnk1, supplied by OriGene, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/recombinant full length wnk1/product/OriGene Average 90 stars, based on 1 article reviews
recombinant full length wnk1 - by Bioz Stars,
2026-04
90/100 stars
|
Buy from Supplier |
|
Proteintech
anti wnk1  Anti Wnk1, supplied by Proteintech, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti wnk1/product/Proteintech Average 93 stars, based on 1 article reviews
anti wnk1 - by Bioz Stars,
2026-04
93/100 stars
|
Buy from Supplier |
|
OriGene
length human wnk1 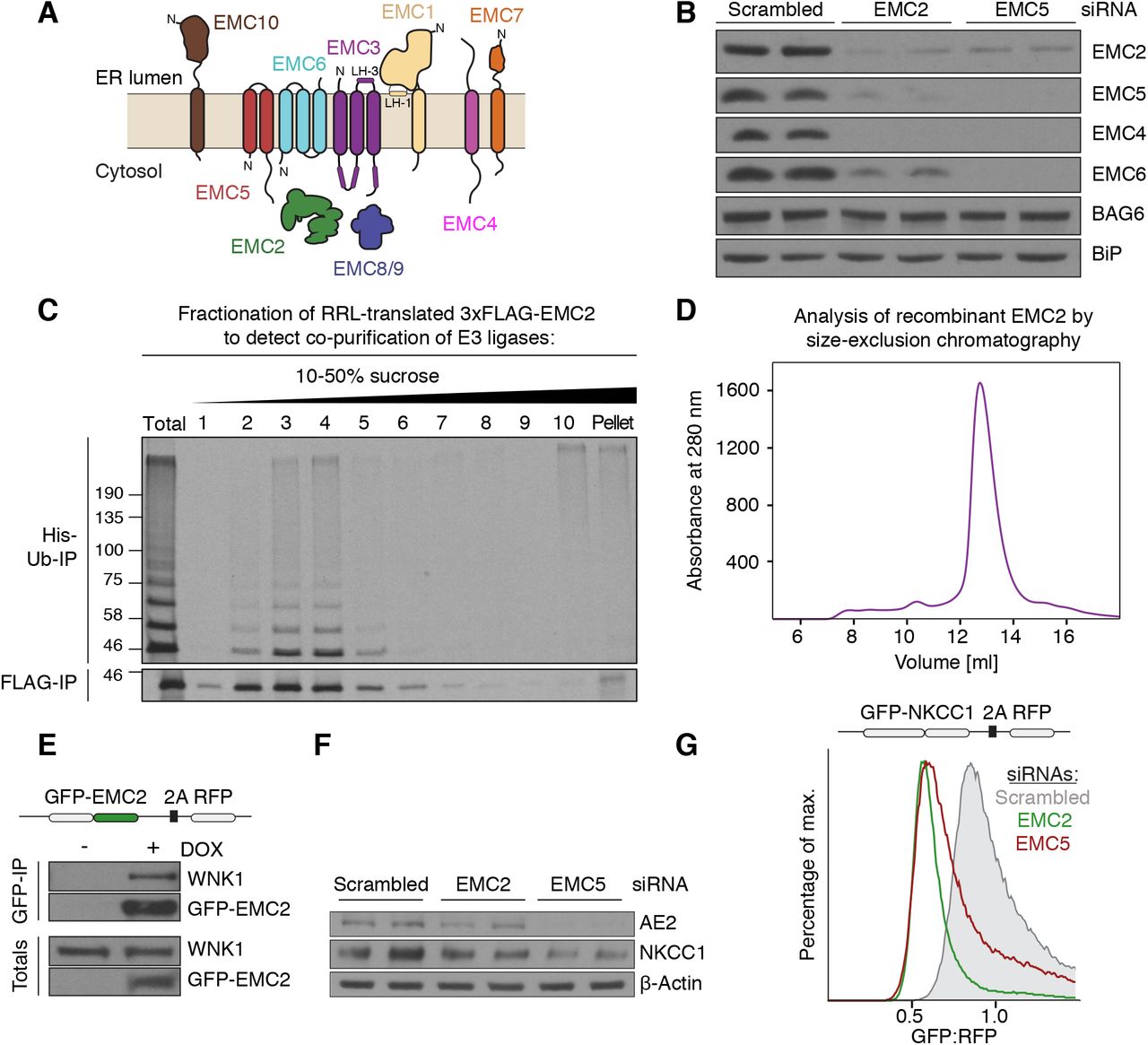 Length Human Wnk1, supplied by OriGene, used in various techniques. Bioz Stars score: 92/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/length human wnk1/product/OriGene Average 92 stars, based on 1 article reviews
length human wnk1 - by Bioz Stars,
2026-04
92/100 stars
|
Buy from Supplier |
|
Carna Inc
wnk1  Wnk1, supplied by Carna Inc, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/wnk1/product/Carna Inc Average 94 stars, based on 1 article reviews
wnk1 - by Bioz Stars,
2026-04
94/100 stars
|
Buy from Supplier |
|
ProSci Incorporated
anti phospho p65  Anti Phospho P65, supplied by ProSci Incorporated, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti phospho p65/product/ProSci Incorporated Average 93 stars, based on 1 article reviews
anti phospho p65 - by Bioz Stars,
2026-04
93/100 stars
|
Buy from Supplier |
|
Alpha Diagnostics
anti-hsn2  Anti Hsn2, supplied by Alpha Diagnostics, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti-hsn2/product/Alpha Diagnostics Average 90 stars, based on 1 article reviews
anti-hsn2 - by Bioz Stars,
2026-04
90/100 stars
|
Buy from Supplier |
|
Carna Inc
flt3 construct  Flt3 Construct, supplied by Carna Inc, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/flt3 construct/product/Carna Inc Average 94 stars, based on 1 article reviews
flt3 construct - by Bioz Stars,
2026-04
94/100 stars
|
Buy from Supplier |
|
OriGene
human wnk1 coding regions  Human Wnk1 Coding Regions, supplied by OriGene, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/human wnk1 coding regions/product/OriGene Average 90 stars, based on 1 article reviews
human wnk1 coding regions - by Bioz Stars,
2026-04
90/100 stars
|
Buy from Supplier |
|
OriGene
hsn2  Hsn2, supplied by OriGene, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/hsn2/product/OriGene Average 90 stars, based on 1 article reviews
hsn2 - by Bioz Stars,
2026-04
90/100 stars
|
Buy from Supplier |
|
OriGene
rat wnk1 mrna  Rat Wnk1 Mrna, supplied by OriGene, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rat wnk1 mrna/product/OriGene Average 90 stars, based on 1 article reviews
rat wnk1 mrna - by Bioz Stars,
2026-04
90/100 stars
|
Buy from Supplier |
Image Search Results
![( A ) Genotyping of Wnk1 -cKO mice mediated by neuron-specific Syn1-Cre . Genomic tail-clip DNA was used for analysis. Lane 1, Wnk1 fl/+ ; Syn1-Cre ; lane 2, Wnk1 fl/fl ; lane 3, Wnk1 fl/fl ; Syn1-Cre . PCR is shown to detect WT vs. Wnk1 fl/fl locus (exon 2 and neo cassette are floxed). PCR forward primer F is located at exon 2. Reverse primers R1 and R2 are located at intron 2 and neo cassette, respectively. Note that Syn1-Cre is only active in neurons, so unexcised Wnk1 fl/fl locus is detected in tail-clip DNA. With large size neo cassette in the floxed locus, the F/R1 primer set does not amplify under the condition of PCR reaction. Additionally, PCR is shown to detect Syn1-Cre using Cre-specific primers. ( B ) Representative Western blot of WNK1 protein in WT and cKO brain regions shown relative to the kidney. Hippo, hippocampus. Cortex, cerebral cortex. ( C ) Quantitation (mean ± SEM) of 4 separate experiments, as shown in B . One WT and cKO mouse for each experiment. WNK1 was normalized to Gapdh and compared with the WT kidney (set as “1”). * P < 0.05, # P < 0.01, KO vs. WT by unpaired t test. ( D ) Atlas of brain section for immunofluorescent staining, as in E and F . OVLT (also known as vascular-organ-of-lamina-terminalis [VOLT]) is marked by a red line. 3V, third ventricle; MnPO, median preoptic nucleus; MPA, medial preoptic area. ( E and F ) Immunofluorescent staining of WNK1 in OVLT neurons colocalized with neuronal marker β-3 tubulin in WT ( E ) and cKO ( F ) mice. Scale bar: 100 μm.](https://pub-med-central-images-cdn.bioz.com/pub_med_central_ids_ending_with_1991/pmc10231991/pmc10231991__jci-133-164222-g062.jpg)
Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A ) Genotyping of Wnk1 -cKO mice mediated by neuron-specific Syn1-Cre . Genomic tail-clip DNA was used for analysis. Lane 1, Wnk1 fl/+ ; Syn1-Cre ; lane 2, Wnk1 fl/fl ; lane 3, Wnk1 fl/fl ; Syn1-Cre . PCR is shown to detect WT vs. Wnk1 fl/fl locus (exon 2 and neo cassette are floxed). PCR forward primer F is located at exon 2. Reverse primers R1 and R2 are located at intron 2 and neo cassette, respectively. Note that Syn1-Cre is only active in neurons, so unexcised Wnk1 fl/fl locus is detected in tail-clip DNA. With large size neo cassette in the floxed locus, the F/R1 primer set does not amplify under the condition of PCR reaction. Additionally, PCR is shown to detect Syn1-Cre using Cre-specific primers. ( B ) Representative Western blot of WNK1 protein in WT and cKO brain regions shown relative to the kidney. Hippo, hippocampus. Cortex, cerebral cortex. ( C ) Quantitation (mean ± SEM) of 4 separate experiments, as shown in B . One WT and cKO mouse for each experiment. WNK1 was normalized to Gapdh and compared with the WT kidney (set as “1”). * P < 0.05, # P < 0.01, KO vs. WT by unpaired t test. ( D ) Atlas of brain section for immunofluorescent staining, as in E and F . OVLT (also known as vascular-organ-of-lamina-terminalis [VOLT]) is marked by a red line. 3V, third ventricle; MnPO, median preoptic nucleus; MPA, medial preoptic area. ( E and F ) Immunofluorescent staining of WNK1 in OVLT neurons colocalized with neuronal marker β-3 tubulin in WT ( E ) and cKO ( F ) mice. Scale bar: 100 μm.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Western Blot, Quantitation Assay, Staining, Marker

Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A ) Water intake, ( B ) urine volume, ( C ) plasma osmolality, ( D ) urine osmolality, ( E ) plasma AVP level, and ( F ) copeptin level of control (Ctrl) and cKO mice at either ad libitum water intake or after 24-hour water restriction (WR). The inset in A shows Western blotting analysis of abundance of total and phospho-WNK1 (p-WNK1) using antibody against total WNK1 and against S382 phospho-WNK1. Arrowheads indicate molecular size 250 kDa. Lysates from WT OVLT tissue at ad libitum water intake and after 24-hour water restriction were immunoprecipitated by anti-WNK1 antibody and probed by anti-WNK1 and anti-p-WNK1 antibody. Representative of 4 separate experiments. Each experiment consists of 1 mouse ad libitum and 1 mouse on water restriction. For statistical analysis was performed with 2-way repeated ANOVA with Šidák post hoc analysis; for statistical analysis of the inset in A , unpaired 2-tailed t test was performed. For bar graphs, n = 6–8 mice, as indicated in scatter plots.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Control, Western Blot, Immunoprecipitation
![( A ) Plasma osmolality, ( B ) [Na + ], ( C ) relative p-WNK1/WNK1 ratio in OVLT, ( D ) plasma AVP, ( E ) urine volume, ( F ) urine osmolality, and ( G ) water intake in WT and Wnk1 -cKO mice after mannitol or vehicle injection. Urine volume and water intake were measured 120 minutes after injection. Other measurements were taken 30 minutes after injection in separate mice from those in which urine and water intake were measured. The inset in C is representative of 3 experiments. Each experiment consists of 1 mouse injected with vehicle and 1 mouse injected with mannitol. Statistical analysis in A , B , and D was performed with 2-way repeated ANOVA with Šidák post hoc analysis; otherwise, unpaired t test was used. For bar graphs in A , B , D – G , n = 5 mice for each experimental condition, as indicated in scatter plot.](https://pub-med-central-images-cdn.bioz.com/pub_med_central_ids_ending_with_1991/pmc10231991/pmc10231991__jci-133-164222-g067.jpg)
Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A ) Plasma osmolality, ( B ) [Na + ], ( C ) relative p-WNK1/WNK1 ratio in OVLT, ( D ) plasma AVP, ( E ) urine volume, ( F ) urine osmolality, and ( G ) water intake in WT and Wnk1 -cKO mice after mannitol or vehicle injection. Urine volume and water intake were measured 120 minutes after injection. Other measurements were taken 30 minutes after injection in separate mice from those in which urine and water intake were measured. The inset in C is representative of 3 experiments. Each experiment consists of 1 mouse injected with vehicle and 1 mouse injected with mannitol. Statistical analysis in A , B , and D was performed with 2-way repeated ANOVA with Šidák post hoc analysis; otherwise, unpaired t test was used. For bar graphs in A , B , D – G , n = 5 mice for each experimental condition, as indicated in scatter plot.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Injection

Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A ) Ruptured whole-cell current-clamp recording for membrane potentials. Pipette and bath solution are indicated. ( B and C ) Membrane potentials of freshly isolated OVLT neurons at baseline, after incubation with 5 mM NaCl for 3 minutes and 5 minutes after washout of 5 mM NaCl hypertonicity. 600 pA currents were injected to depolarize membrane potential from the resting potential –55 mV to +150 mV. B and C represent examples of NaCl-responsive and nonresponsive neurons, respectively. ( D ) Treatment with pan-WNK kinase inhibitor (WNK463). Green and cyan bars indicate responsive (R) and nonresponsive (NR), respectively. WNK463 treatment significantly decreased the percentage distribution of responsive neurons vs. vehicle (Veh) treatment. P < 0.01, WNK463 vs. Veh, by 2-tailed Fisher’s exact test. ( E ) Wnk1 -cKO eliminated NaCl responsiveness. P < 0.01, cKO vs. WT, by 2-tailed Fisher’s exact test. In D and E , OVLT neurons were isolated form 4–5 mice for vehicle-treated, WNK463-treated, WT, and cKO groups.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Membrane, Transferring, Isolation, Incubation, Injection

Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A ) Injection of AAV-retro-Cre virus into PVN of tdTomato-EGFP reporter mice resulted in green fluorescence in neurons of OVLT nuclei, which otherwise exhibited tomato red fluorescence. Scale bar: 200 μm. ( B ) PVN injection of AAV-retro-Cre virus into Wnk1 fl/fl mice resulted in deletion of Wnk1 in OVLT compared with control experiments with injection of AAV-retro-Cre virus into PVN of WT mice. Scale bar: 100 μm. ( C ) Urine volume, ( D ) urine osmolality, and ( E ) plasma osmolality of Wnk1 fl/fl mice before and after injection with AAV-retro-Cre virus during at libitum and after water restriction (WR). ( F ) Urine volume, ( G ) urine osmolality, and ( H ) plasma osmolality of WT mice before and after injection with AAV-retro-Cre virus. Data shown are mean ± SEM from before injection (labeled retro-AAV –) and after injection (labeled retro-AAV +). Statistical analysis by 2-way repeated ANOVA with Šidák post hoc analysis. n = 4–6 mice as indicated by scatter plots.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Injection, Virus, Fluorescence, Control, Labeling

Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A and B ) Cooopeptin release in Wnk1 fl/fl and control WT mice with PVN injected with AAV-retro-Cre virus. Statistical comparison was made by paired t test between ad libitum and WR. ( C and D ) In separate groups of experimental ( Wnk1 fl/fl ) and control (WT) mice, OVLT neurons were isolated for recording of membrane potential oscillation. Pie charts show distribution of neurons that exhibit membrane potential oscillation responsive and nonresponsive to HTS (5 mM NaCl). P < 0.01 between pie chart in C and D by 2-tailed Fisher’s exact test. In A and B , n = 5 Wnk1 fl/fl and WT mice per experiment, as indicated in scatter plots.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Control, Injection, Virus, Comparison, Isolation, Membrane

Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: WT mice or mice heterozygous for GOF Cl – -insensitive Wnk1 -knockin ( Wnk1 -KI) allele received AAV-Cre virus injection in OVLT. ( A ) Relative abundance of phospho-OSR/SPAK (p-OSR/SPAK) in KI mice before (–) and after (+) injection, as measured by Western blotting analysis of OVLT using antibody against S373-phospho-SPAK/S325-phospho-OSR1. The inset shows representative Western blotting of 3 separate experiments. Each experiment consists of 3 replicates of WT and 3 Wnk1 -KI mice. Each data point in the bar graph represents the average of 3 replicates. Statistical analysis by unpaired t test. ( B ) Plasma AVP level, ( C ) urine volume, ( D ) urine osmolality in heterozygous Wnk1 -KI mice in which OVLT was injected with AAV-Cre virus, ( E ) plasma AVP level, ( F ) urine volume, and ( G ) urine osmolality of WT mice in which OVLT was injected with AAV-Cre virus. In B – G , n = 5 mice, as indicated in line plots. Statistical analysis by paired t test.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Knock-In, Virus, Injection, Western Blot

Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A ) Schematic of the virus-mediated KO of Wnk1 in PVN-projecting OVLT neurons via injection of Cre-expressing retrograde virus at the PVN region. ( B ) Representative coronal section of the mouse brain injected with Cre-expressing virus at the PVN region. Scale bar: 1 mm. ( C ) Overlay of epifluorescence and IR-DIC images showing Cre-expressing neurons in the OVLT region. Scale bar: 10 μm. A recording pipette attached to a Cre-expressing cell was illustrated. ( D ) Top: Representative traces of spontaneous firing recorded from a NaCl-R neuron (R; cyan trace) and a NaCl-NR neuron (NR; red trace) in the WT mice. Bottom: Histogram of z score from the representative NaCl-R and NaCl-NR cells. ( E ) Distribution of the Δ z score in response to 5 mM NaCl stimulation of all recorded neurons in WT mice. The dashed line indicates 0.5. ( F ) Pie chart showing distribution of NaCl-R and NaCl-NR PVN-projecting OVLT neurons in WT mice. ( G ) Top: Representative traces of spontaneous firing recorded from a NaCl-R neuron and a NaCl-NR neuron in the Wnk1 –conditional KO (cKO) mice. Bottom: Histogram of z score from the representative NaCl-R and NaCl-NR cells. ( H ) Distribution of the Δ z score in response to 5 mM NaCl stimulation of all recorded neurons in Wnk1 -cKO mice. The dashed line indicates 0.5. ( I ) Pie chart showing distribution of NaCl-R and NaCl-NR PVN-projecting OVLT neurons in Wnk1 -cKO mice. * P = 0.032, between F and I , 2-tailed Fisher’s exact test. The WT group consists of recordings of 22 cells from 13 mice; the cKO group consists of 24 cells from 11 mice.
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Virus, Injection, Expressing, Transferring

Journal: The Journal of Clinical Investigation
Article Title: WNK1 promotes water homeostasis by acting as a central osmolality sensor for arginine vasopressin release
doi: 10.1172/JCI164222
Figure Lengend Snippet: ( A ) WNK1 exists in conformational equilibrium between chloride-bound autoinhibited dimer and chloride-free activation-competent monomer. Hyperosmolality extracts water from the cell and from the catalytic core of WNK1, which facilitates chloride unbinding, allowing autophosphorylation at S382 and be activated ( – ). WNK1 may activate Kv3.1 directly or indirectly through other intermediaries such as OSR1/SPAK. ( B ) Kv3.1 is a high-threshold voltage-gated K + channel activated by membrane depolarization to –20 mV or above ( , ). Activation of Kv3.1 shortens action potential duration, increases after hyperpolarization (AHP), and thus increases firing frequency (illustrated by red trace). Conversely, inhibition of Kv3.1 decreases firing frequency (blue trace). In support of this notion, we have found that TEA increased the action potential half-width (data not shown). ( C ) Exponential curvilinear relationship between AVP release and plasma osmolality begins at the threshold of approximately 280 mOsm/kg. WNK1 activation by cellular dehydration (Excitatory pathway; thick green line) plays an important role in AVP release by hyperosmolality. Additional mechanism(s) may be involved, at least for secretion at the basal state, which may include tonic inhibition of osmosensory neurons (Inhibitory pathway; thick solid red line). Loss of hypotonicity-mediated inhibitory pathway (thick dotted red line) may also contribute to hyperosmolality-induced AVP release. Compensation by the additional pathways may account for apparent similar AVP release defects in OVLT-selective deletion of WNK1 (by direct shRNA injection) versus neuronal deletion of WNK1. Extracellular hypertonicity may also activate WNK1 signaling cascade through molecular crowding of the protein (ref. ) (data not shown).
Article Snippet: The following primary and secondary antibodies were used: anti-WNK1 (NB600-225 and AF2849, Novus Biologicals); anti-β3 tubulin (MAB1195, R&D Systems);
Techniques: Activation Assay, Membrane, Inhibition, shRNA, Injection

Journal: bioRxiv
Article Title: Compensatory ion transport buffers daily protein rhythms to regulate osmotic balance and cellular physiology
doi: 10.1101/2020.05.28.118398
Figure Lengend Snippet: (a) Schematic of the WNK/OXSR1 pathway and regulation of the N(K)CC and KCC transporters. (b) Kinase activity assays for WNK1 and 3-Phosphoinositide Dependent Protein Kinase 1 (PDPK1) upon increasing concentrations of NaCl or polyethylene glycol (PEG). WNK1 but not PDPK1 is sensitive to increased macromolecular crowding (mimicked by PEG). Note that WNK1 is inhibited by high concentrations of Cl − . ( c ) Phosphorylation of cellular OXSR1 upon hypotonic treatment, indicating that decreased intracellular Cl − increases OXSR1 phosphorylation. The addition of a phosphatase on the cell lysates confirms the identity of P-OXSR1. ( d ) Representative immunoblots and quantification showing OXSR1 and phospho-OXSR1 abundance in fibroblasts sampled every 3 h for 3 days in constant conditions (n=3). The asterisk indicates OXSR1 band. p-value indicates comparison of damped cosine wave with straight-line fit (null hypothesis = no rhythm). ( e ) Soluble protein abundance at peak and trough of protein rhythms in fibroblasts ± 30 μM of OXSR1 inhibitor closantel (n=6). Data were normalised to control at 24 h. Statistical test used was two-way ANOVA with Sidak’s MCT. Mean ± SEM shown throughout.
Article Snippet:
Techniques: Activity Assay, Western Blot

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) Topology of the EMC subunits based on the structure of the human EMC . Note that EMC8 and 9 are paralogs and their binding to EMC2, and thus incorporation into the EMC, is mutually exclusive. ( B ) The stability of soluble and membrane-embedded EMC subunits is interdependent in cells. HeLa cells were treated with scrambled, EMC2, or EMC5 siRNA and harvested for Western blotting with the indicated antibodies. Similar results were observed in HEK293T, U2OS, and RPE1 cell lines and has been reported in previous studies ( ; ). ( C ) Soluble EMC2 stably recruits an E3 ubiquitin ligase. An in vitro translation reaction of 35 S-methionine-labeled 3xFLAG-EMC2 was fractionated on a 10-50% (w/v) sucrose gradient. Unfractionated translation (Total) and all fractions were subjected to FLAG-IP and subsequently incubated with ATP, His-tagged ubiquitin (His-Ub), as well as E1 and E2 enzymes to detect the presence of co-purifying E3 ligase activity. Ubiquitinated species were enriched after elution from the FLAG resin by Ni 2+ -chelate affinity chromatography. ( D ) E. coli -purified EMC2 is a monodisperse, soluble protein. His 14 - bd NEDD8-tagged EMC2 was purified on a Superdex 200 Increase 10/300 size-exclusion chromatography column. UV absorbance at 280 nm was monitored throughout the purification. ( E ) WNK1 co-purifies with EMC2 from human cells. HEK293T cell lines stably expressing GFP-EMC2 were used for purification of GFP-tagged EMC2 and its interacting proteins under native conditions using a biotinylated Avi-SUMO Eu1 -tagged anti-GFP nanobody (see methods). Samples of total lysate and eluate were analyzed by Western blot with anti-GFP and anti-WNK1 antibodies. ( F ) Endogenous NKCC1 expression relies on a functional EMC. HeLa cells were treated with either scrambled, EMC2, or EMC5 siRNA and harvested for Western blotting with the indicated antibodies. Expression of endogenous AE2, a previously identified EMC substrate, displays a similar dependence on the EMC for expression. ( G ) NKCC1 is post-translationally destabilized in the absence of the EMC. HEK293T cells stably expressing GFP-NKCC1-2A-RFP were treated with either scrambled, EMC2, or EMC5 siRNA and the GFP:RFP ratio was analyzed by flow cytometry. The observed post-translational destabilization of NKCC1 upon EMC depletion is consistent with a requirement for EMC in NKCC1 biogenesis.
Article Snippet: The coding sequence of full
Techniques: Binding Assay, Western Blot, Stable Transfection, In Vitro, Labeling, Incubation, Activity Assay, Affinity Chromatography, Purification, Size-exclusion Chromatography, Expressing, Functional Assay, Flow Cytometry

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) HEK293T cells stably expressing GFP-EMC2 were transiently transfected with plasmids encoding either BFP or BFP-tagged siRNA-resistant WNK1. Cells were treated with either scrambled or WNK1 siRNA, and BFP-positive cells were analyzed by flow cytometry. The relative levels of GFP-EMC2, normalized to an internal RFP expression control (described in methods), are plotted as a histogram and reflect changes in subunit stability. ( B-D ) Scrambled or WNK1 siRNA treatment of EMC4-GFP, RFP-squalene synthase (SQS; a tail-anchored EMC substrate) and RFP-VAMP2 (a tail-anchored WRB-CAML substrate) cell lines and analysis by flow cytometry.
Article Snippet: The coding sequence of full
Techniques: Stable Transfection, Expressing, Transfection, Flow Cytometry

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) HEK293T cells stably expressing GFP-EMC2-2A-RFP were treated with a scrambled control or EMC5 siRNA, or three independent WNK1 siRNAs for 72 hours. 24 hours following siRNA transfection the fluorescent reporter was induced for 48 hours. Cells were then analyzed by flow cytometry and their GFP:RFP ratio plotted as a histogram. ( B ) HEK293T cell lines stably expressing (left) EMC4-GFP-2A-RFP, and (right) EMC5-GFP-2A-RFP, were harvested 48 hours after reporter induction with doxycycline (DOX), solubilized in 1% (w/v) LMNG, and the GFP-tagged subunits purified using an anti-GFP nanobody (Nb) (see methods) . The eluates were analyzed by SDS-PAGE and Sypro Ruby staining. GFP-tagged EMC4 and 5 are incorporated into the intact EMC and thus co-purify endogenous EMC subunits, which were identified by mass spectrometry. The characterization of the stable HEK293T GFP-EMC2-2A-RFP cell line was described before . ( C+D ) Stable HEK293T cell lines expressing either ( C , left) EMC5-GFP-2A-RFP or ( C , right) GFP-2A-RFP without any fusion partner, ( D , left) TRAM2-GFP-2A-RFP or ( D , right) OPRK1-GFP-2A-RFP were treated with the indicated siRNAs for 72 hours. 24 hours following siRNA transfection the fluorescent reporter was induced for 48 hours with DOX. Cells were then analyzed by flow cytometry and their GFP:RFP ratio plotted.
Article Snippet: The coding sequence of full
Techniques: Stable Transfection, Expressing, Transfection, Flow Cytometry, Purification, SDS Page, Staining, Mass Spectrometry

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) Stable HEK293T cell lines expressing either GFP-EMC2-2A-RFP (left), GFP-2A-RFP-SQS (middle) or GFP-2A-RFP-VAMP2 (right) were treated either with DMSO or 1 μ M pan-WNK inhibitor WNK463 (IC 50 = 5 nM for WNK1) for 68 hours. 20 hours following drug treatment the fluorescent reporter was induced for 48 hours with DOX. Cells were then analyzed by flow cytometry and their GFP:RFP ratio plotted as a histogram. No effect of WNK463 on EMC2 stability or EMC activity was observed. ( B ) Left: HEK293T cells were treated with either scrambled or WNK1 siRNA for 72 hours. Right: HEK293T cells were treated with DMSO or 1 μ M WNK463 for 68 hours. Cells were then harvested for Western blotting with the indicated antibodies. Note that WNK463 treatment reduced SPAK/OSR1 S-motif phosphorylation (pSer373 in SPAK, pSer325 in OSR1) much like a WNK1 knockdown.
Article Snippet: The coding sequence of full
Techniques: Expressing, Flow Cytometry, Activity Assay, Western Blot

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) FLAG-tagged WNK1 and its truncations were tested for binding to EMC2 (see methods). The EMC2 binding site was mapped to a previously uncharacterized C-terminal peptide that is predicted to form a conserved amphipathic α-helix (see ). Polar residues are shown in blue and hydrophobic residues in yellow. ( B ) HEK293T cells stably expressing GFP-EMC2 were transiently transfected with plasmids encoding either BFP or BFP-tagged wild type (wt) or L2250K mutant (mut) WNK1 helix. Cells were treated with either scrambled or WNK1 siRNA and analyzed by flow cytometry as in . ( C ) Intact EMC, purified from stable EMC5-3xFLAG HEK293T cells, and recombinant EMC2 were incubated with 3xHA-tagged WNK1 helix and immunoprecipitated with anti-HA resin. Totals and eluates were analyzed by Western blotting with the indicated antibodies.
Article Snippet: The coding sequence of full
Techniques: Binding Assay, Stable Transfection, Expressing, Transfection, Mutagenesis, Flow Cytometry, Purification, Recombinant, Incubation, Immunoprecipitation, Western Blot

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) High sequence conservation of the WNK1 helix in vertebrates. Top: Shown are sequence alignments corresponding to residues 2241-2266 in human WNK1. The following species were included: Human ( Homo sapiens ), Mouse ( Mus musculus ), Dog ( Canis lupus familiaris ), Frog ( Xenopus tropicalis ), Alligator ( Alligator sinensis ), Chicken ( Gallus gallus ), Fish ( Nothobranchius furzeri ) and Shark ( Callorhinchus milii ). Bottom: Alignment of human WNK1, WNK2 and WNK3 in the region corresponding to the amphipathic α-helix (residues 2241-2266 in human WNK1). WNK4 has no region with significant sequence homology. ( B ) The equivalent helical regions in WNK2 (residues 2153-2178) and WNK3 (residues 1641-1666) also interact with EMC2. Purified 3xFLAG-tagged EMC2 was incubated with either buffer or His 14 - bd SUMO-tagged WNK helix variants and then subjected to FLAG-IP. Bound proteins were eluted with SDS sample buffer and analyzed by SDS-PAGE and Coomassie staining. ( C ) The hydrophobic face of the WNK1 amphipathic helix interacts with EMC2. Purified 3xFLAG-tagged EMC2 was incubated with either buffer or His 14 - bd SUMO-tagged WNK1 helix variants and then subjected to FLAG-IP. Bound proteins were eluted with SDS sample buffer. Samples of totals, unbound and elution were analyzed by SDS-PAGE and Coomassie staining.
Article Snippet: The coding sequence of full
Techniques: Sequencing, Purification, Incubation, SDS Page, Staining

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) HEK293T cells stably expressing GFP-EMC2 were transfected with plasmids encoding BFP, BFP-tagged WNK1 wild type (wt) or L2250K mutant helix. Cells were analyzed by flow cytometry and their relative GFP intensity, normalized to an internal RFP expression control, was plotted against BFP intensity. ( B ) An 35 S-methionine-labeled representative hydrophobic protein (SEC61β) was produced with the BpA photo–crosslinker within the TMD as a complex with calmodulin (CaM) in the PURE in vitro translation system. The CaM-SEC61β complexes were incubated with EMC2 alone or pre-formed complexes of EMC2 with increasing concentrations of purified wild type (wt) or L2250K mutant WNK1 helix. Except for the -UV control, all reactions were irradiated and analyzed by SDS-PAGE and autoradiography. ( C ) 35 S-methionine labeled wild type EMC2, or the indicated point mutants were translated in RRL and tested for binding to 3xFLAG-tagged WNK1, EMC8 or EMC3 via co-immunoprecipitation using anti-FLAG resin. EMC2 mutants that are unable to bind WNK1 also do not bind EMC8, but are capable of interacting with EMC3, indicating proper folding. ( D ) Depicted is the surface representation of EMC2 (PDB 6WW7; ) in which hydrophobic residues have been highlighted in yellow (left) or mutations that affected WNK1 binding in purple (right). The WNK1 binding site overlapped with the binding site of EMC8 (blue), which is characterized by an extended hydrophobic patch. EMC2 is viewed from the cytosol. ( E ) 35 S-methionine labeled EMC2 was translated in RRL and purified either directly via a 3xFLAG-tag or indirectly via 3xFLAG-tagged WNK1 helix or EMC8 (translation of 3xHA-tagged EMC2). The purified complexes were analyzed for ubiquitination competence to detect the presence of co-purifying E3 ubiquitin ligases. Following enrichment for ubiquitinated species, the total intensity of ubiquitinated species in all samples was quantified and normalized to the 3xFLAG-EMC2 sample.
Article Snippet: The coding sequence of full
Techniques: Stable Transfection, Expressing, Transfection, Mutagenesis, Flow Cytometry, Labeling, Produced, In Vitro, Incubation, Purification, Irradiation, SDS Page, Autoradiography, Binding Assay, Immunoprecipitation

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) EMC2 and EMC8 form a complex in human cells. Stable HEK293T cell lines were induced with DOX to express GFP-tagged EMC2 or EMC8. After mechanical lysis under non-solubilizing conditions, GFP-tagged EMC2 or EMC8 and their interacting proteins were purified under native conditions from lysate using a biotinylated anti-GFP nanobody (see methods). Samples of total lysate and eluate were blotted and probed with the indicated antibodies. ( B+C ) WNK1 helix binding to EMC2 is incompatible with EMC8 interaction. ( B ) Purified 3xFLAG-tagged EMC2 was incubated either with purified EMC8, WNK1 helix, or both and then subjected to FLAG-IP. Totals and eluates were analyzed by SDS-PAGE and Coomassie staining. WNK1 helix binding to EMC2 is mutually exclusive to binding of EMC8. ( C ) Stoichiometric amounts of EMC2, EMC8 and WNK1 helix were incubated to allow complex formation and then analyzed by size-exclusion chromatography. Selected fractions were analyzed by SDS-PAGE and Coomassie staining. Note that EMC8 outcompetes WNK1 helix binding to EMC2 (compare with ). ( D ) In yeast, the analogous WNK1/EMC8 interface on EMC2 is occupied by the N-terminus of EMC4. A structural overlay of yeast EMC2 (gray: PDB 6WB9) and human EMC2 (green: PDB 6WW7) is shown with or without human EMC8 (blue) and yeast EMC4 N-terminus (pink) ( ; ). The N-terminus of EMC4 is poorly conserved, consistent with its non-essential role in binding EMC2 in metazoans.
Article Snippet: The coding sequence of full
Techniques: Lysis, Purification, Binding Assay, Incubation, SDS Page, Staining, Size-exclusion Chromatography

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) WNK1 helix binding to EMC2 is compatible with EMC3 interaction. Purified EMC2 was incubated with EMC3 coiled coil (3-CC), EMC3 C-terminus (3-CT), and WNK1 helix, and the resulting complexes were analyzed by size-exclusion chromatography. Peak fractions were analyzed by SDS-PAGE and Coomassie staining. ( B ) The EMC2-WNK1 complex is not capable of binding to the EMC5 C-terminus (5-CT). As in , using purified EMC2 and 5-CT, but with WNK1 helix instead of EMC8. ( C ) In the absence of an EMC8 homolog in yeast, S. cerevisae ( sc ) EMC2 binds sc EMC5 directly. Full-length sc EMC5-3xFLAG was translated alone or co-translated with 3xHA-tagged sc EMC2 in RRL in the presence of cRMs and subjected to FLAG-IP as described in . Bound proteins were eluted with 3xFLAG peptide and analyzed alongside samples of total translation by SDS-PAGE and autoradiography. ( D ) Comparison of the yeast (PDB 6WB9) and human EMC2-5 interfaces (PDB 6WW7) ( ; ). In contrast to human EMC2, the N-terminus of yeast EMC2 packs against an elongated unstructured loop of yeast EMC5, resulting in a more extensive interface. Interface residues in human EMC2 and EMC5 that were mutated in (G) are highlighted. ( E ) EMC2 strongly accumulates upon proteasome inhibitor treatment. HEK293T cells stably expressing GFP-EMC2-2A-RFP were induced with DOX and treated with either DMSO or 20 nM proteasome inhibitor bortezomib. Cells were were analyzed by flow cytometry and their GFP:RFP ratio plotted as a histogram. ( F ) Unassembled EMC2 preferentially engages WNK1 in human cells. Cells were treated as in (E) and then lysed mechanically under non-solubilizing conditions, GFP-tagged EMC2 and its interacting proteins were purified from lysate with biotinylated anti-GFP nanobody. Normalized samples of total lysate and eluate were analyzed by Western blotting with the indicated antibodies. Following bortezomib treatment WNK1 co-purification with EMC2 increases, whereas EMC8 co-purification decreases. This indicates that under conditions where EMC8 becomes limiting, EMC2 accumulates in complex with WNK1. ( G ) 35 S-methionine-labeled 3xFLAG-tagged EMC5 (or the indicated mutants) was translated in the presence of 3xHA-tagged EMC2 (or the indicated mutants) and EMC8 in RRL in the presence of canine rough microsomes (cRMs). cRMs were isolated by centrifugation through a sucrose cushion, resuspended, and solubilized in digitonin. The resulting detergent lysate was subjected to FLAG-IP and analyzed by SDS-PAGE and autoradiography. ( H ) As in G. EMC2 HLY is a triple mutant of H189E, L190E and Y191K that was previously described . ( I ) As in , HEK293T cells stably expressing exogenous 3xFLAG-BFP-tagged wild type or the HLY mutant EMC2 were lysed and subjected to native FLAG-IP. Eluates were analyzed by Western blotting.
Article Snippet: The coding sequence of full
Techniques: Binding Assay, Purification, Incubation, Size-exclusion Chromatography, SDS Page, Staining, Autoradiography, Stable Transfection, Expressing, Flow Cytometry, Western Blot, Copurification, Labeling, Isolation, Centrifugation, Mutagenesis

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: WNK1 stabilizes unassembled EMC2 by using a conserved C-terminal amphipathic α-helix to prevent nonspecific binding and premature degradation in the cytosol. EMC8 then displaces WNK1 in the cytosol, allowing assembly with the membrane-bound subunits EMC3 and EMC5.
Article Snippet: The coding sequence of full
Techniques: Binding Assay

Journal: bioRxiv
Article Title: Regulated assembly of the ER membrane protein complex
doi: 10.1101/2020.07.20.213066
Figure Lengend Snippet: ( A ) WNK1 co-immunoprecipitates with vesicle proteins. Stable HEK293T cell lines expressing 3xFLAG-tagged full length WNK1 wild type (wt) or WNK1 helix mutant (mut) (L2250K, L2253K, W2257K) were lysed mechanically under non-solubilizing conditions. WNK1 and its interacting proteins were immunoprecipitated from lysate with anti-FLAG resin. After elution with 3xFLAG peptide, the eluates were analyzed by SDS-PAGE and Sypro Ruby staining. ( B ) Eluates from (A) were analyzed by Western blotting with the indicated antibodies. Mass spectrometry revealed Clathrin and Clathrin-associated vesicle proteins as the highest specific hits. Importantly, these interactions are sensitive to detergents commonly used for cell lysis (e.g. 1% Triton-X-100 or NP-40), explaining why they were not observed before . Mutation of WNK1’s amphipathic helix disrupts its interaction with vesicle-resident proteins. ( C ) HEK293T cell lines stably expressing full-length 3xFLAG-WNK1(wt) or WNK1 (mut = L2250K, L2253K, W2257K) were either left untreated (isotonic) or stimulated with 0.2 M Sorbitol for 5 min prior to fixation. Cells were then stained with anti-FLAG and Clathrin antibodies via indirect immunofluorescence and analyzed by confocal microscopy.
Article Snippet: The coding sequence of full
Techniques: Expressing, Mutagenesis, Immunoprecipitation, SDS Page, Staining, Western Blot, Mass Spectrometry, Lysis, Stable Transfection, Immunofluorescence, Confocal Microscopy